Your Content Goes Here
List of peer-reviewed project publications:
Microbiome 12┃29 July 2024┃Article number: 141 (2024)┃https://doi.org/10.1186/s40168-024-01858-1
Authors: Arno R. Bourgonje (UMCG), Nicolai V. Hörstke (MUW), Michaela Fehringer (MUW), Gabriel Innocenti (MUW) & Thomas Vogl (MUW/PI)
Abstract:
- Background
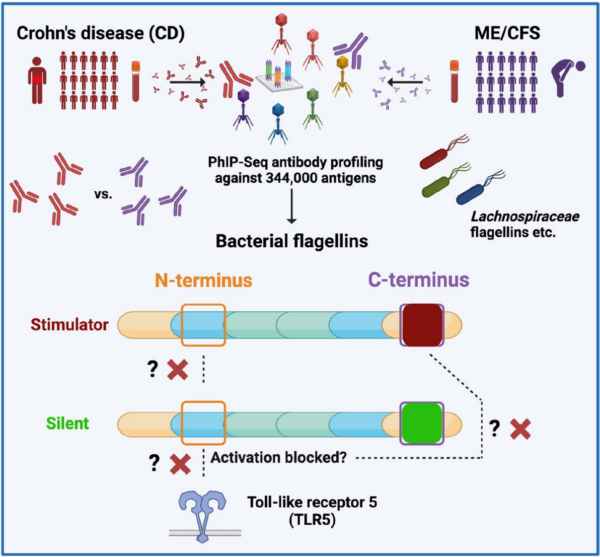
Elevated systemic antibody responses against gut microbiota flagellins are observed in both Crohn’s disease (CD) and myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), suggesting potential serological biomarkers for diagnosis. However, flagellin-specific antibody repertoires and functional roles in the diseases remain incompletely understood. Bacterial flagellins can be categorized into three types depending on their interaction with toll-like receptor 5 (TLR5): (1) “stimulator” and (2) “silent” flagellins, which bind TLR5 through a conserved N-terminal motif, with only stimulators activating TLR5 (involving a C-terminal domain); (3) “evader” flagellins of pathogens, which entirely circumvent TLR5 activation via mutations in the N-terminal TLR5 binding motif. - Results
Here, we show that both CD and ME/CFS patients exhibit elevated antibody responses against distinct regions of flagellins compared to healthy individuals. N-terminal binding to Lachnospiraceae flagellins was comparable in both diseases, while C-terminal binding was more prevalent in CD. N-terminal antibody-bound flagellin sequences were similar across CD and ME/CFS, resembling “stimulator” and “silent” flagellins more than evaders. However, C-terminal antibody-bound flagellins showed a higher resemblance to the stimulator than to silent flagellins in CD, which was not observed in ME/CFS. - Conclusions
These findings suggest that antibody binding to the N-terminal domain of stimulator and silent flagellins may impact TLR5 activation in both CD and ME/CFS patients. Blocking this interaction could lead commensal bacteria to be recognized as pathogenic evaders, potentially contributing to dysregulation in both diseases. Furthermore, elevated antibody binding to the C-terminal domain of stimulator flagellins in CD may explain pathophysiological differences between the diseases. Overall, these results highlight the diagnostic potential of these antibody responses and lay a foundation for deeper mechanistic studies of flagellin/TLR5 interactions and their impact on innate/adaptive immunity balance.
Graphical Abstract:
PNAS┃May 9, 2024┃121 (23) e2309674121┃https://doi.org/10.1073/pnas.2309674121
Authors: Balázs Koncz, Gergő Mihály Balogh, Máté Manczinger (BRC)
Abstract: The identification of immunogenic peptides has become essential in an increasing number of fields in immunology, ranging from tumor immunotherapy to vaccine development. The nature of the adaptive immune response is shaped by the similarity between foreign and self-protein sequences, a concept extensively applied in numerous studies. Can we precisely define the degree of similarity to self? Furthermore, do we accurately define immune self? In the current work, we aim to unravel the conceptual and mechanistic vagueness hindering the assessment of self-similarity. Accordingly, we demonstrate the remarkably low consistency among commonly employed measures and highlight potential avenues for future research.


